Traditional refrigerators and air conditioners rely on vapor-compression cooling technology, which suffers from high energy consumption. To address this issue, scientists and engineers around the world are striving to find better alternatives, and solid-state phase-change cooling technology has emerged as one of the most promising solutions.
The key to this technology lies in a unique property of solid materials: when subjected to different external fields, such as magnetic, electric, or pressure fields, the internal structure of the material, known as its “phase,” undergoes a change. This transformation absorbs or releases heat, thereby achieving cooling. All the existing methods share a common limitation: their cooling effect only occurs within a very narrow temperature range around the “phase transition temperature” (typically about ±10 Kelvin,). To achieve a broader temperature span, many materials with different phase transition temperatures must be combined—like stringing beads on a skewer—into a multi-stage cooling device, which poses a major challenge for scientists.

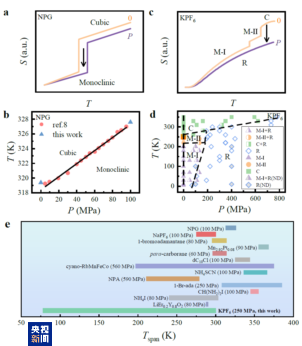
Recently, our team has made a significant breakthrough in addressing this challenge. For the first time, we observed the “all-temperature-range barocaloric effect” in an inorganic plastic crystal material called potassium hexafluorophosphate (KPF₆). By applying pressure, KPF₆ can generate a cooling effect across an exceptionally wide temperature range—from room temperature (around 25 °C) down to the cryogenic regions of liquid nitrogen (−196 °C), liquid hydrogen (−253 °C), and even liquid helium (−269 °C), making it the only solid-state phase-change cooling material to date that operates effectively across the entire temperature spectrum.
At room temperature and ambient pressure, KPF₆ has a face-centered cubic structure in which the PF₆ molecular groups can freely and randomly rotate. As the temperature decreases or pressure increases, it undergoes two structural transitions (phase changes), transforming into different monoclinic structures, which in turn gives rise to strong endothermic or exothermic effects.
This research was published on August 20 (Beijing time) in Nature Communications, paving the way for the development of a new generation of efficient and environmentally friendly all-solid-state cooling technologies.
Comparison of Conventional and All-Temperature-Range Barocaloric Effects
This work has been covered by prominent media outlets, including CCTV News and ScienceNet.

