A research team has developed a Pd₂ dual-atom catalyst that achieves both high activity and high selectivity in acetylene semihydrogenation, overcoming a long-standing challenge in the purification of ethylene feedstock for the polyolefin industry.
Ethylene, a commodity chemical with an annual production exceeding 200 million tons, is primarily produced by naphtha steam cracking. The resulting ethylene-rich gas stream contains trace amounts of acetylene (0.5–3%), which poisons the Ziegler-Natta catalysts used in downstream ethylene polymerization, reducing catalyst lifetime and product quality. Industrially, acetylene is removed via semihydrogenation to ethylene. However, the thermodynamic advantage of overhydrogenation to ethane creates a fundamental trade-off: catalysts with high activity often suffer from low ethylene selectivity, while selective catalysts typically exhibit low activity.
A collaborative team led by Prof. LIU Hongyang from the Institute of Metal Research, Chinese Academy of Sciences, together with collaborators, has now broken this trade-off. Using a solvent-mediated dispersion strategy, they successfully anchored atomically dispersed Pd₂ dual-atom sites on a defect-rich graphene support (Pd₂/ND@G).
The Pd₂ dual-atom catalyst achieved complete acetylene conversion at 100 °C with an ethylene selectivity of 93.2%, and maintained stable performance over 100 hours of testing. Compared to Pd single-atom catalysts, the turnover frequency increased from 0.151 s⁻¹ to 1.953 s⁻¹—a 13-fold enhancement—without compromising selectivity.
Synchrotron radiation-based characterizations, temperature-programmed experiments, H₂-D₂ exchange reactions, isotope labeling, and density functional theory calculations revealed the mechanism. Unlike single atoms, the Pd₂ dual-atom site exhibits enhanced metallicity and strong synergy between the two palladium atoms, enabling efficient co-activation of both acetylene and hydrogen while maintaining weak physical adsorption of ethylene—similar to that on single-atom sites. This unique adsorption behavior effectively breaks the activity-selectivity trade-off.
The work, published in Nature Communications, provides a new strategy for designing low-cost, highly efficient sub-nanometer atomic-scale hydrogenation catalysts with broad implications for the polyolefin industry.
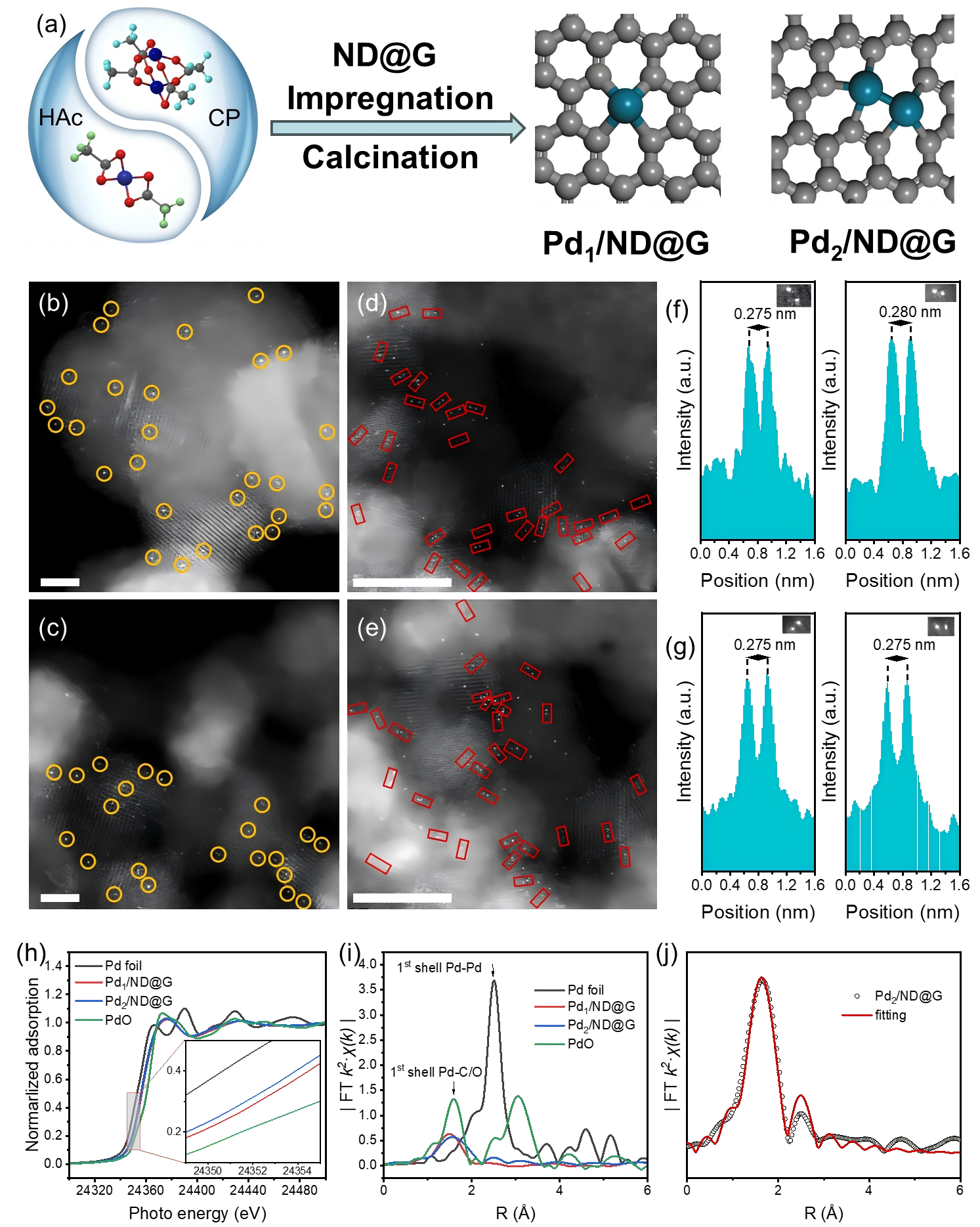
Catalyst structural characterization. (a) Synthesis schematic, (b-f) HAADF-STEM images and corresponding line intensity profiles, (g) Pd K-edge XANES spectra, (h-i) Pd EXAFS spectra and their fitting. (Image by IMR)
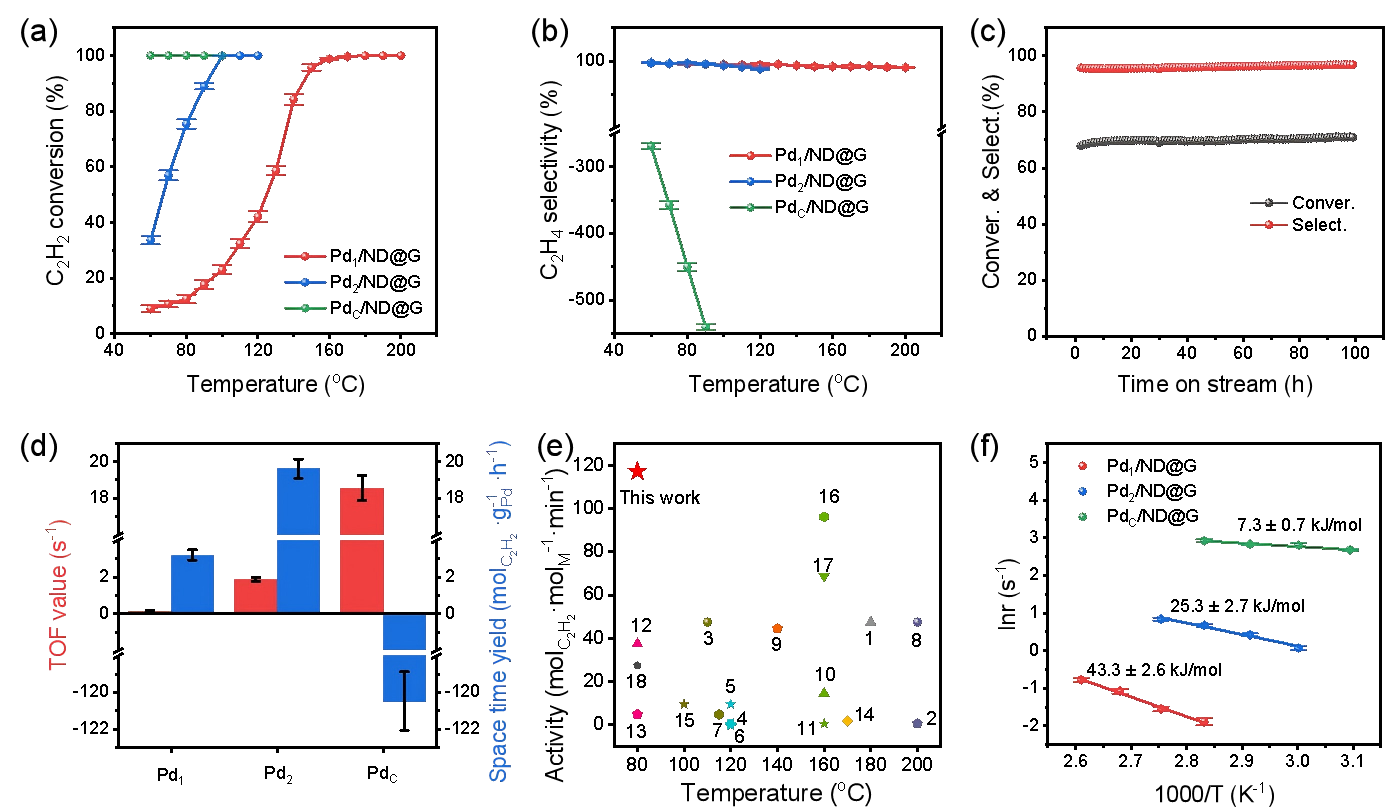
Catalytic performance for acetylene semihydrogenation. (a) Catalytic activity, (b) ethylene selectivity, (c) long-term stability test, (d) turnover frequency (TOF) and space-time yield (STY), (e) comparison of specific activity, (f) apparent activation energy. (Image by IMR)
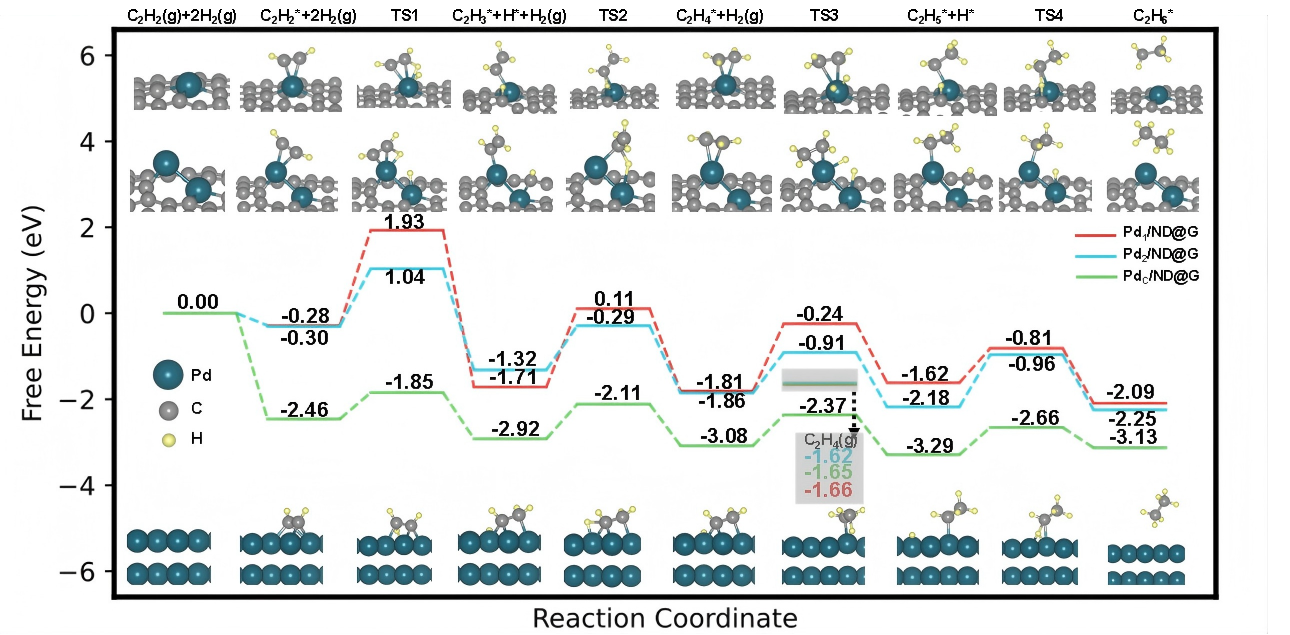
Density functional theory (DFT) calculations. (Image by IMR)
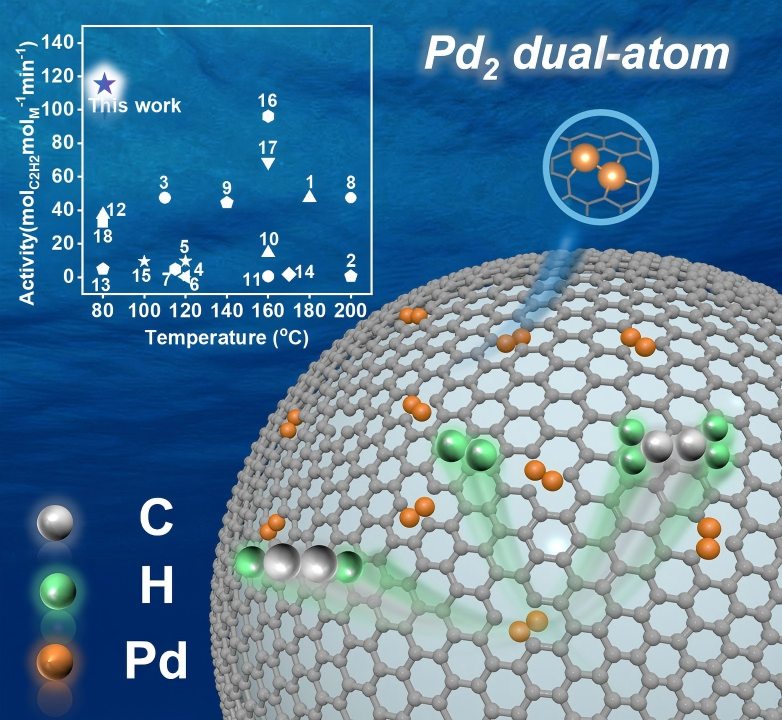
Schematic illustration of acetylene semihydrogenation catalyzed by Pd₂/ND@G. (Image by IMR)