A research team has developed a novel iron-complex negative electrolyte that enables an alkaline all-iron flow battery to operate for over 6,000 cycles without capacity decay, addressing a long-standing bottleneck in low-cost, long-duration energy storage for grid-scale renewable energy integration.
As the world accelerates its transition to renewable energy, the need for low-cost, long-life, and intrinsically safe long-duration energy storage has become critical. Alkaline all-iron flow batteries are an ideal candidate due to the abundance of iron, inherent safety, and decoupled power and capacity. However, conventional iron-based negative electrolytes suffer from poor electrochemical reversibility, easy decomposition of active species, and ligand crossover through membranes, severely limiting cycle life and practical applications.
Researchers Prof. TANG Ao and Prof. LI Ying from the Shenyang National Laboratory for Materials Science at the Institute of Metal Research, Chinese Academy of Sciences, have now overcome these bottlenecks. They proposed a synergistic design strategy combining high steric hindrance with a negatively charged interface, successfully developing an ultra-stable iron complex negative electrolyte. The results were published in Advanced Energy Materials.
Starting from molecular design, the team synthesized 11 iron complexes based on 12 organic ligands rich in hydroxyl and sulfonic acid groups. Through theoretical calculations and multiple rounds of screening, they identified the bis-ligand chelate complex [Fe(HPF)BHS]⁴⁻ as the optimal candidate. This complex features a bulky steric structure formed by multidentate strong coordination, while the sulfonate and hydroxyl groups create a negatively charged interface. This dual protection, via Donnan exclusion, effectively prevents hydroxide ions from attacking the iron center—thereby avoiding active species decomposition—and significantly reduces the crossover of both active species and free ligands across the membrane, fundamentally solving the problems of electrolyte cross-contamination and degradation.
Performance tests demonstrated comprehensive breakthroughs. At 80 mA·cm⁻², the battery operated for over 6,000 cycles with no capacity decay and an average coulombic efficiency of 99.4%. At a high current density of 150 mA·cm⁻², the energy efficiency remained 78.5%, with a peak power density of 392.1 mW·cm⁻². Even at a high concentration of 0.9 M, stable cycling for 2,000 cycles was achieved with an energy efficiency of 71.5%. Multiscale characterization confirmed that [Fe(HPF)BHS]⁴⁻ reshapes the solvation structure of iron ions, reducing ligand crossover by two orders of magnitude compared to conventional systems.
This work establishes a new design principle for iron-based electrolytes, greatly enhancing the lifetime and cost-effectiveness of all-iron flow batteries and offering a promising solution for large-scale energy storage to support carbon neutrality and energy security.
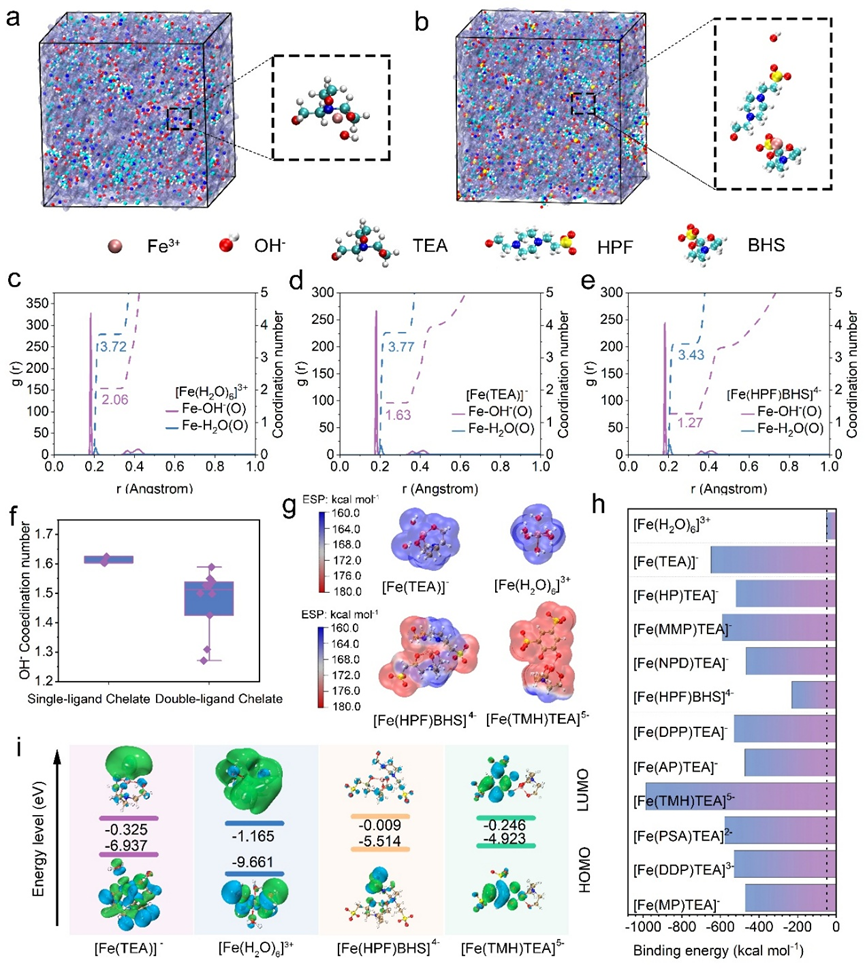
Design and screening strategy for electrolyte active materials in alkaline all-iron flow batteries. (Image by IMR)
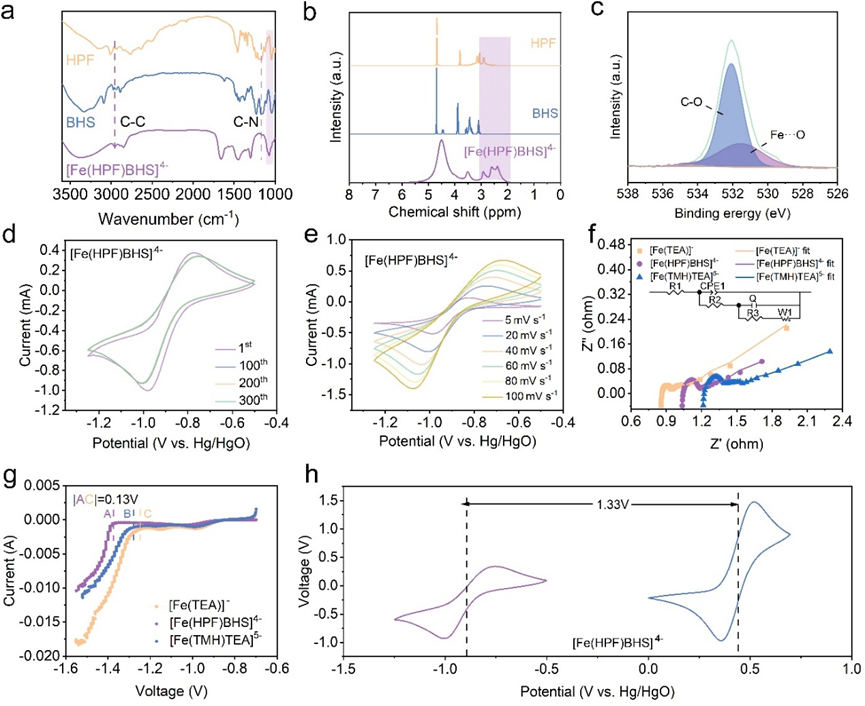
Electrochemical performance of the negative electrolyte active material for alkaline all-iron flow batteries. (Image by IMR)
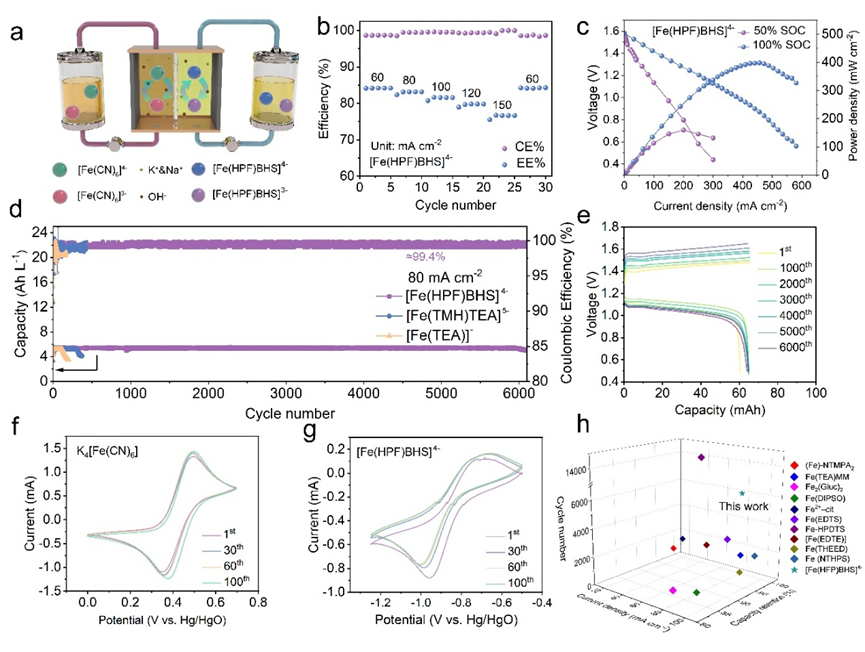
Cell performance of the alkaline all-iron flow battery. (Image by IMR)
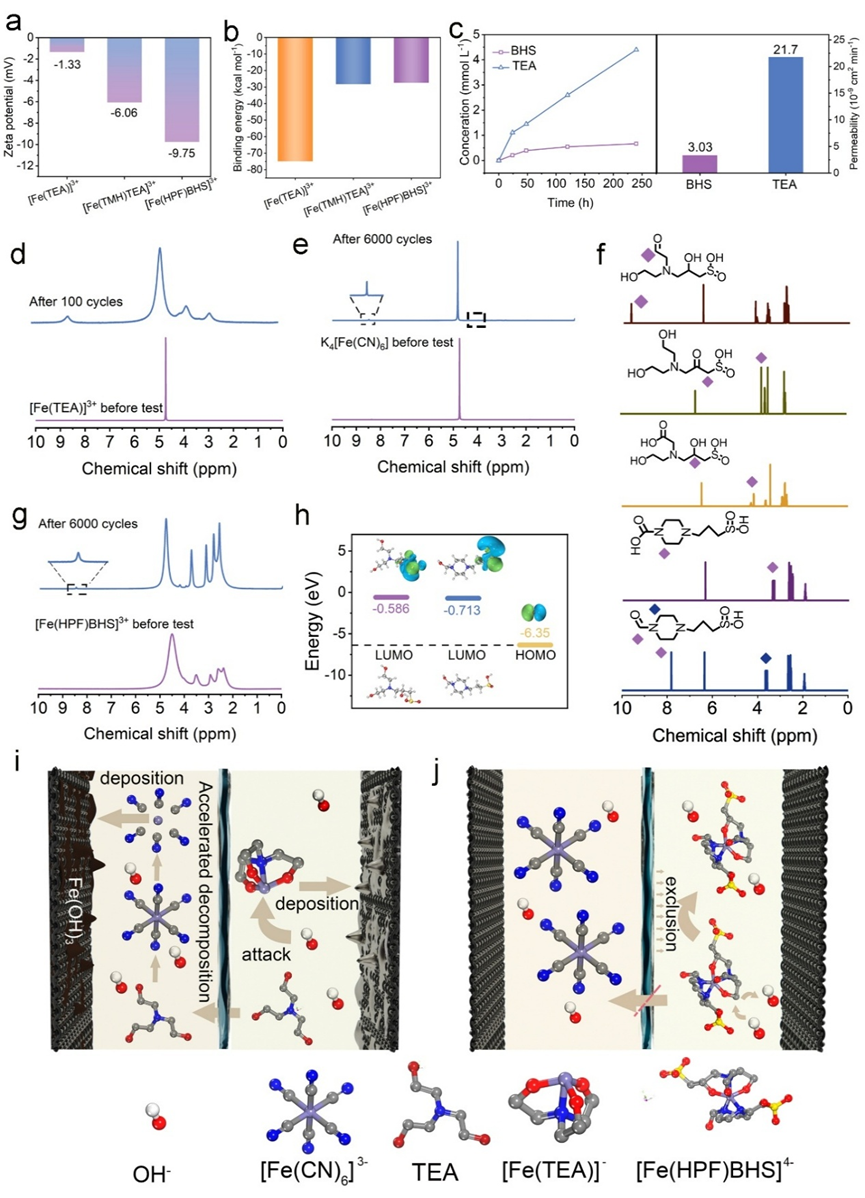
Failure mechanism of the [Fe(HPF)BHS]⁴⁻ negative electrolyte active material. (Image by IMR)