A research team from the Institute of Metal Research, Chinese Academy of Sciences, has established a new criterion for designing electrocatalysts for solid-phase conversion reactions in high-energy lithium batteries, challenging the conventional thermodynamics-based framework.
Lithium–sulfur and lithium–oxygen batteries are promising candidates for next-generation energy storage due to their ultra-high theoretical energy densities. However, their practical application has been hindered by the sluggish and incomplete conversion of insulating solid intermediates, such as Li2S2 and Li₂O₂. Unlike gas- or liquid-phase reactions, these solid intermediates continuously deposit on the electrode/catalyst surface during battery cycling, blocking electron and ion transport pathways and leading to premature reaction termination. Traditional catalyst design, relying on thermodynamic descriptors like adsorption energy, fails to capture the critical role of electron transport in solid-state conversion systems.
The team, led by Prof. LI Feng, Prof. SUN Zhenhua and Academician. CHENG Huiming, conducted large-scale density functional theory calculations on 351 dual-atom catalysts and proposed an electronic property criterion that emphasizes efficient ion and electron conduction as essential for high catalytic activity. Their findings reveal that during the initial reaction stage, thermodynamic energy barriers dominate the nucleation kinetics of Li2S2. However, as insulating solid intermediates accumulate, electron transport rapidly becomes the rate-determining step. The proportion of electrons near the Fermi level shows a much stronger correlation with catalytic activity than thermodynamic energy barriers in the mid-to-late stages of solid-phase conversion.
Based on this insight, the team designed a homonuclear cobalt–cobalt dual-atom catalyst (DA-CoCo). This catalyst significantly enhances the conductivity of solid intermediates through effective orbital coupling, transferring active catalytic sites from the passivated catalyst surface to the conductive intermediate surface and enabling efficient ion and electron transport. An ampere-hour-scale lithium–sulfur pouch cell incorporating DA-CoCo achieved a specific energy of 459 Wh kg-¹, validating the new design principle in practical complex systems.
The work, published in Nature Catalysis, provides a universal design principle for electrochemical systems limited by insulating solid-phase conversion and opens a new direction for electrocatalyst design.
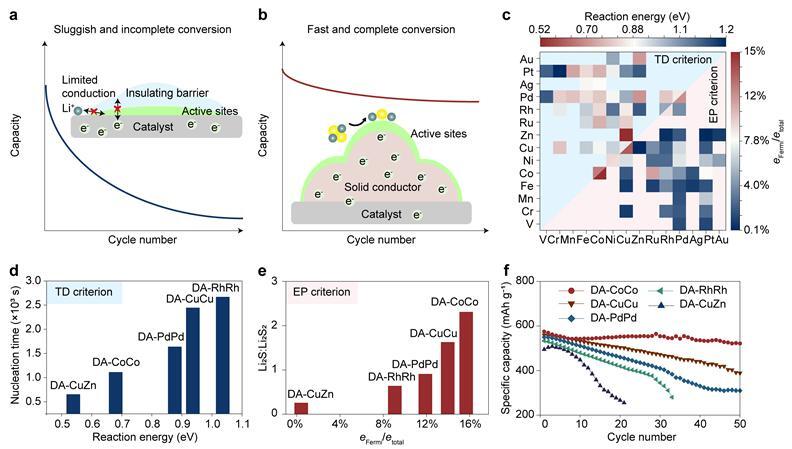
Role of thermodynamic properties of catalytic materials and electronic properties of products in solid-phase conversion reactions of lithium–sulfur batteries. (Image by IMR)
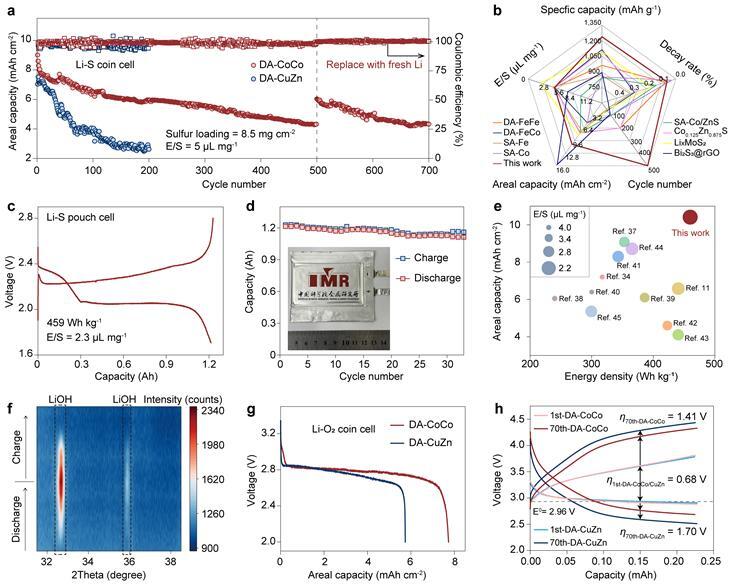
Performance enhancement of lithium–sulfur and lithium–air batteries enabled by dual-atom catalytic materials. (Image by IMR)