A collaborative research team has developed a heteronuclear Rh-Co dual-atom catalyst that enables highly efficient nitrile hydrogenation under mild conditions, breaking the long-standing activity-selectivity trade-off in this important industrial reaction. These findings were published in Nature Communication.
Secondary amines are widely present in pharmaceuticals, agrochemicals and functional materials, with global annual amine production exceeding six million tonnes. Catalytic hydrogenation of nitriles is a key route to these valuable compounds. However, traditional noble metal catalysts suffer either from poor selectivity (typically 60–75% towards the desired secondary amine) due to overhydrogenation or coupling side reactions, or require harsh conditions (temperatures >373 K and H₂ pressures of 3–10 MPa). While single-atom catalysts offer enhanced selectivity through isolated active sites, a single metal center struggles to co-activate both H₂ and bulky nitrile substrates, leading to insufficient catalytic activity.
To address this challenge, researchers from the Institute of Metal Research, Chinese Academy of Sciences, Peking University, and Chongqing University constructed an atomically dispersed heteronuclear Rh-Co dual-atom catalyst supported on defective graphene (Rh1Co1/ND@G). Using a stepwise anchoring strategy, they first immobilized Rh atoms on the support, followed by the introduction of Co(NH3)4²+ complexes and hydrogen reduction, successfully forming Rh-Co atomic pairs with an average interatomic distance of approximately 2.55 Å. Electronic structure analysis further revealed strong electronic coupling between the two metal species.
The Rh₁Co₁/ND@G catalyst achieved exceptional performance in the hydrogenation of benzonitrile to dibenzylamine under mild conditions (333 K, 0.6 MPa H₂). A turnover frequency of 4068 h⁻¹ was recorded, 1.6-fold higher than that of the Rh single-atom counterpart, while the selectivity toward dibenzylamine exceeded 98%. These figures surpass those of all previously reported heterogeneous catalysts for this reaction. The catalyst also demonstrated excellent durability, maintaining over 85% product yield after 12 recycling runs. Moreover, it showed broad substrate tolerance, efficiently converting various electron-rich or electron-deficient aromatic nitriles, heterocyclic nitriles and even acetonitrile into the corresponding secondary amines in high yields.
Mechanistic studies combining kinetic experiments, temperature-programmed desorption, in situ infrared spectroscopy, and density functional theory calculations elucidated the synergistic catalytic mechanism. The Rh site primarily activates and dissociates molecular hydrogen, while the Co site enhances benzonitrile adsorption, particularly through π-interactions with the aromatic ring. The cooperative interaction polarizes the C≡N bond and lowers the activation energy barrier of the rate-determining step from 0.81 eV on the Rh single-atom catalyst to 0.61 eV on the Rh-Co dual-atom catalyst, thereby kinetically promoting the reaction at lower temperature.
This work provides a new strategy for designing high-performance atomically dispersed hydrogenation catalysts for green chemistry and fine chemical synthesis.
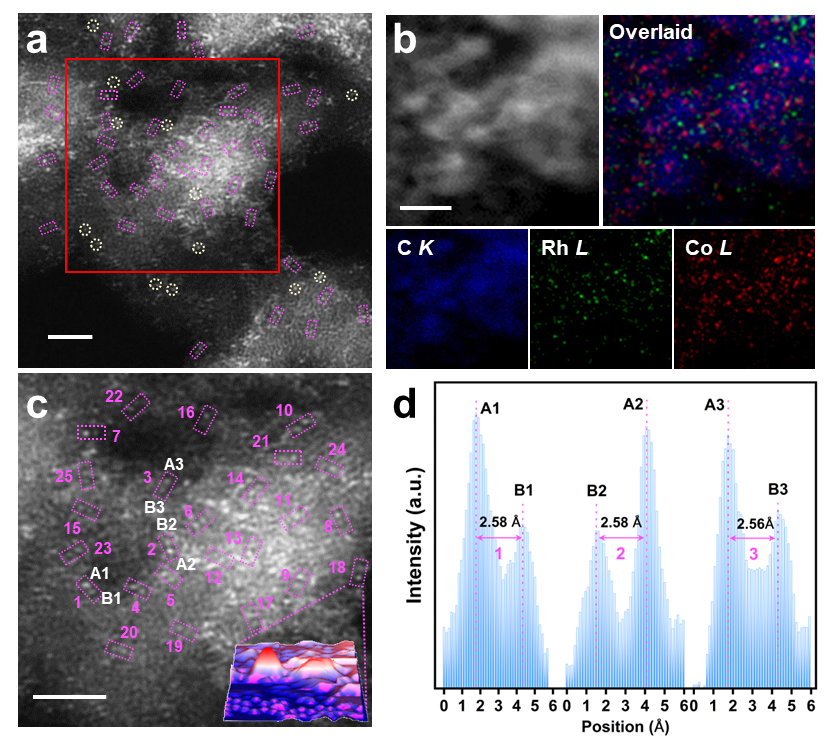
Catalyst structural characterization. (a‑c) HAADF-STEM images, (d) interatomic distance distribution. (Image by IMR)
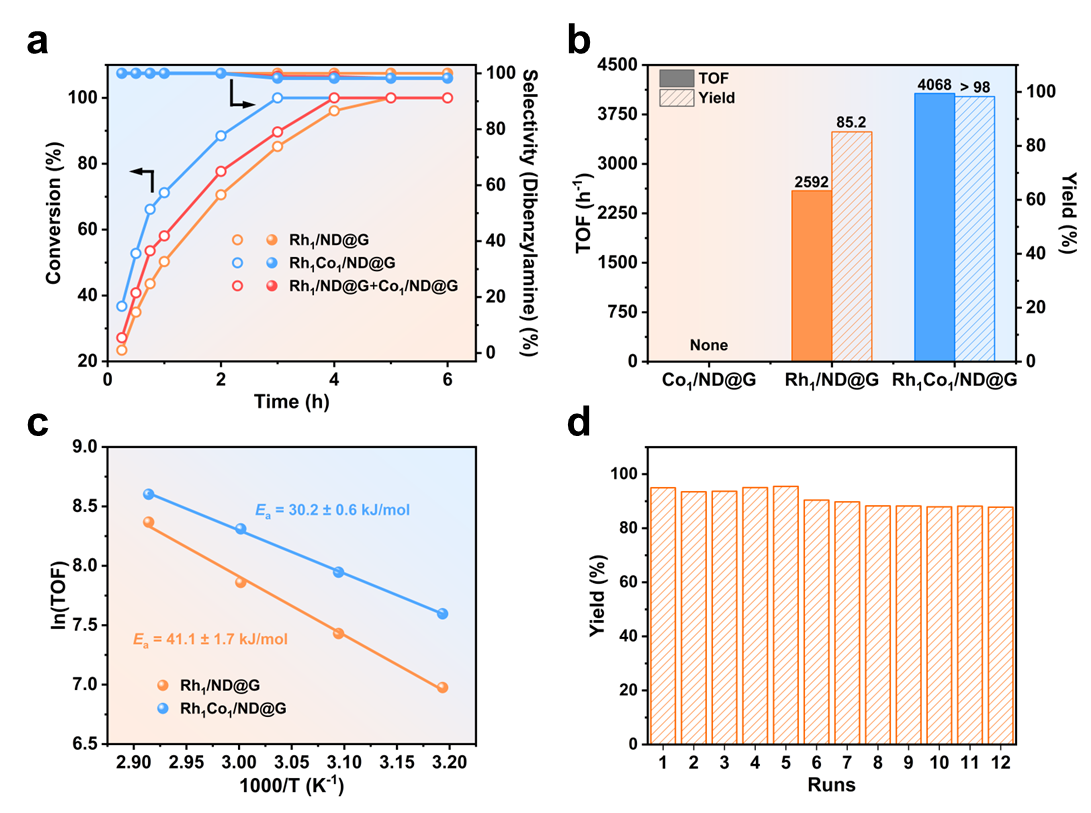
Catalytic performance of benzonitrile hydrogenation. (a) Time-conversion profiles, (b) TOF comparison, (c) apparent activation energy, (d) recycling stability. (Image by IMR)
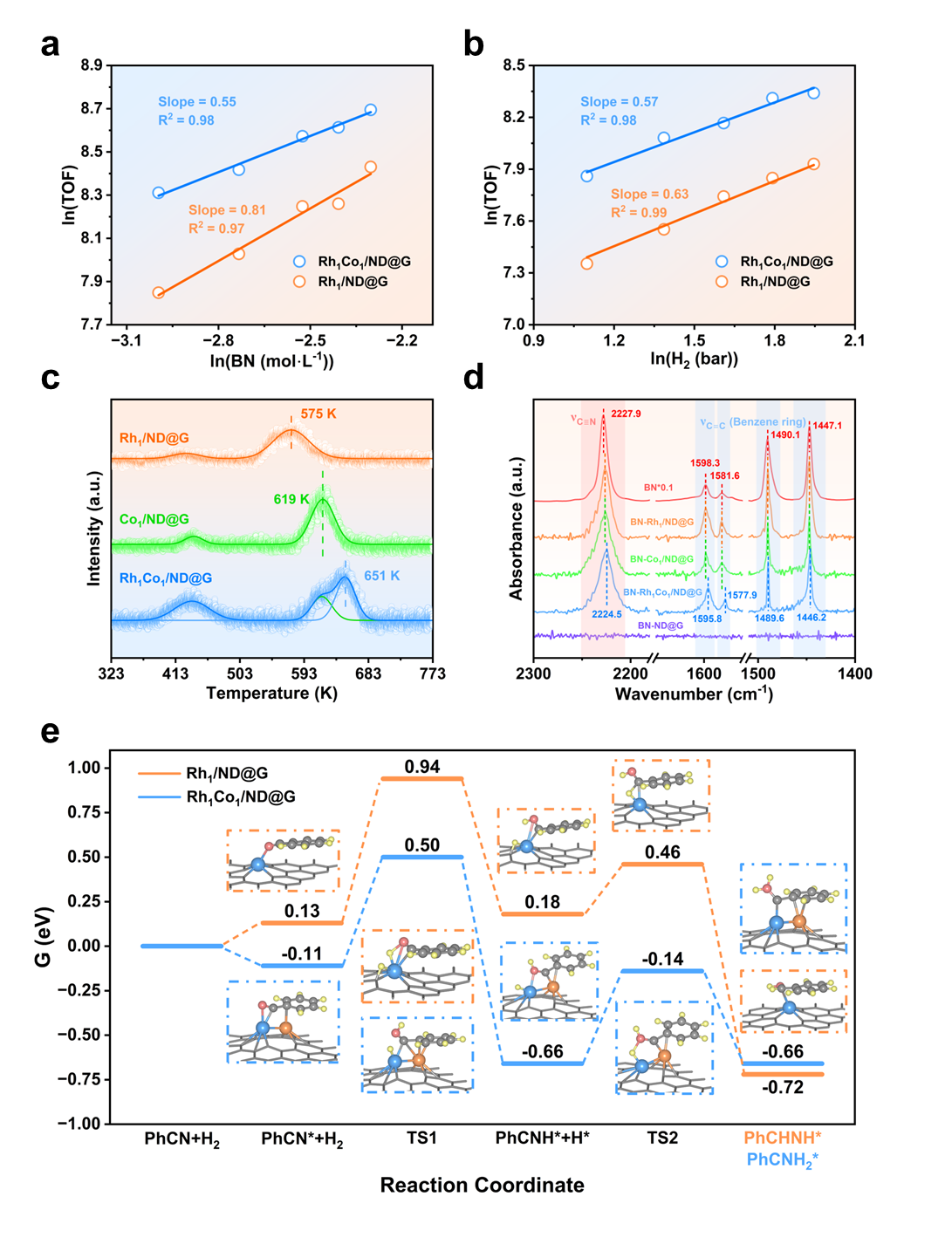
Mechanistic studies. (a‑b) Kinetic studies, (c) BN-TPD, (c) BN in situ FTIR spectra, (e) DFT calculations. (Image by IMR)

Schematic illustration of benzonitrile hydrogenation catalyzed by Rh₁Co₁/ND@G. (Image by IMR)